Abstract
Background
Sickle cell disease (SCD) and thalassemia are inherited hemoglobinopathies characterized by lifelong anemia. In SCD, a single mutation in the β-globin gene results in sickle hemoglobin (HbS) that polymerizes upon deoxygenation, causing red blood cells (RBCs) to sickle leading to a variety of complications. In thalassemia, mutation(s) in α- or β-globin genes result in reduced or absent adult Hb causing ineffective erythropoiesis and downstream complications. The resultant anemias are exacerbated by impaired RBC function due to decreased ATP content. Supportive care and agents like hydroxyurea are used most in SCD, with a subset of patients (pts) on regular transfusions. Regular or episodic transfusions, with their own set of complications, are the mainstay of treatment for thalassemias.
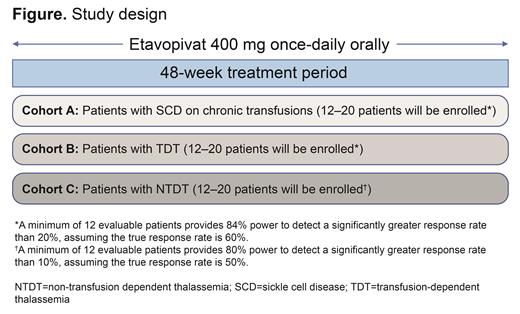
Etavopivat, a potent, selective, oral, small molecule activator of erythrocyte pyruvate kinase (PKR) increases ATP and decreases 2,3 diphosphoglycerate (2,3-DPG). In a Phase 1 study, etavopivat 300‒600 mg once daily in pts with SCD not regularly transfused was well-tolerated, improved hematological markers, decreased hemolysis, and improved markers of RBC functional health (Brown et al. EHA 2021 # EP1202). Etavopivat 200 and 400 mg once daily (dose levels predicted to provide the desired pharmacodynamic response profiles) is being evaluated in a Phase 2/3 study of pts with SCD who are not on chronic transfusions (The Hibiscus Study, NCT04624659). Herein we describe the design of a Phase 2, open-label, multicenter study (NCT04987489) evaluating the efficacy and safety of etavopivat in pts with: SCD on chronic transfusions (Cohort A), transfusion-dependent thalassemia (Cohort B), and non-transfusion-dependent thalassemia (Cohort C).
Study Design and Methods
Up to 20 pts aged 12-65 years will be enrolled in each of the three cohorts described above. Pts will receive etavopivat 400 mg once daily for 48-wks (Figure).
In cohorts A and B, pts must have received ≥6 RBC units in the 24 wks before the first dose of etavopivat without a >35-day transfusion-free period during that period and be on iron chelation therapy for >3 months before enrollment. Additionally, pts in Cohort A should have received ≥24 monthly transfusions for the prevention or treatment of primary stroke. Pts in Cohort C should have a Hb ≤10 g/dL. Key exclusion criteria include significant infection, hepatic/renal dysfunction, history of malignancy/cardiac/pulmonary disease, a drug malabsorption disorder, prior/concomitant therapies ≤3 months before the first dose (eg, chronic systemic glucocorticoids, new chelation therapy).
Baseline assessments will include medical, disease, transfusion, and medication histories. Transfusions received during the study (every ~3-5 wks) will be recorded and include Hb values before and ≥15 minutes after transfusion, number of RBC units, and volume of packed RBCs. If a pt has an increase in pre-transfusion Hb of ≥1.0 g/dL compared with their baseline pre-transfusion Hb, the Investigator may delay transfusion by 1 wk or reduce the number of RBC units transfused. In pts with SCD, RBC exchange may also be performed.
The primary endpoint for Cohorts A/B is erythroid response defined as the proportion of pts with ≥20% reduction in transfusions over a continuous 12-wk treatment period versus baseline, and for Cohort C is Hb response at Wk 12 defined as an increase in Hb of ≥1.0 g/dL from baseline. For Cohorts A/B, secondary and exploratory endpoints include the proportion of pts with a reduction in transfusions over 12 wks of ≥33% and ≥50%, respectively, and a reduction in transfusions over 12, 24, and 48 wks; and for Cohort C, Hb response at Wks 24 and 48, and changes from baseline in Hb over 12, 24, and 48 wks. The following additional endpoints will be assessed for all cohorts: changes from baseline in quality of life as assessed via the Short Form Health Survey and Patient-Reported Outcome Measurement Information System Fatigue Scale; changes from baseline in the levels of serum ferritin at 12, 24, and 48 wks; liver iron at 48 wks; 2,3-DPG and ATP; pharmacokinetics; and safety. All primary endpoints will be analyzed using a 1-sided test at α=0.025.
Summary
Etavopivat is a novel, investigational, PKR activator designed to improve the functional health of RBCs. This Phase 2 study will assess the safety of etavopivat and its impact on Hb levels and transfusion burden in pts with SCD or thalassemia.
Lal: Chiesi: Consultancy; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; bluebird bio, Inc.: Research Funding; Protagonist Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Agios Pharmaceuticals: Consultancy; Insight Magnetics: Research Funding; Novartis: Research Funding; Terumo Corporations: Research Funding; La Jolla Pharmaceutical Company: Research Funding. Brown: Novo Nordisk: Consultancy; Novartis: Consultancy, Research Funding; Imara: Consultancy, Research Funding; Forma Therapeutics: Research Funding; Pfizer: Research Funding; Global Blood Therapeutics: Consultancy, Research Funding. Coates: Apo Pharma: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Bluebird Bio: Consultancy, Membership on an entity's Board of Directors or advisory committees; Agios: Consultancy, Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Chiesi: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Consultancy, Honoraria, Research Funding; Forma Pharma: Consultancy; Sangamo: Consultancy; UpToDate: Patents & Royalties; Vifor Pharma: Consultancy; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees. Kalfa: Agios Pharmaceuticals, Inc.: Other: Steering Committee, Research Funding; FORMA Therapeutics, Inc: Research Funding. Kwiatkowski: Bioverativ: Research Funding; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees; Agios Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; CRISPR: Research Funding; Imara: Consultancy, Research Funding; Silence Therapeutics: Consultancy; Sangamo: Research Funding; bluebird bio: Consultancy, Research Funding; Chiesi: Research Funding; Vertex: Research Funding. Brevard: Forma Therapeutics, Inc.: Current Employment, Current equity holder in publicly-traded company, Current holder of stock options in a privately-held company. Potter: Forma Therapeutics, Inc: Current Employment, Current equity holder in publicly-traded company; Bristol Myers Squibb: Ended employment in the past 24 months. Wood: Forma Therapeutics, Inc: Current Employment, Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months. Sheth: Bluebird bio: Consultancy; CRISPR: Consultancy; Bristol Myers Squibb: Consultancy, Research Funding; Imara: Research Funding; Chiesi: Consultancy; Agios: Consultancy; Dispersol: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal